Amination
Amines are incredibly useful intermediates and they are a key functionality of most biologically active molecules.
In modern organic synthesis practice, the most common reactions to form C-N bonds, besides the synthesis of amides (LINK TO SPECIFIC SECTION), are the nucleophilic substitution of alkyl halides and the Buchwald-Hartwig amination of aryl halides. Another one is reductive amination of carbonyls, which will be discussed in a separate section (not among top 5 reactions).
Amination of alkyl halides
Often referred to as alkylation of amines, the nucleophilic substitution of alkyl halides with aliphatic or heteroaromatic amines is a “historical” organic reaction. It’s relatively simple, well established reaction and still a primary method for the alkylation of amines with aliphatic compounds.
At the contrary, the aromatic nucleophilic substitution has been almost completely abandoned in favour of milder, more robust and efficient catalytic methods, in particular the Hartwig-Buchwald C-N coupling.

The amination of alkyl halides can be run in several different solvents, with DMF and THF being the most common ones and occurs in the presence of a base, such as Na2CO3, or tertiary amines. The temperature is a key parameter to affect the reaction kinetics and while the substitution can happen at room temperature, reflux conditions are widely reported for less reactive building blocks. The alkylation of heteroaromatic amines often requires the use of strong bases, such as sodium bis(trimethylsilyl)amide and cryogenic conditions.
The rate of alkylation follows the order primary amine > secondary amine > tertiary amine and the reactivity of the halide derivative follows the electronegativity of the halide substituent. Bromide and iodides are the most practical building blocks, while chlorides remain fairly common for their broad commercial availability. The use of NaI is a practical way to generate in-situ a more reactive alkyl iodide if the starting material is a different halide.
The nucleophilic substitution of alkyl halides is not considered a very green reaction as it produces a halide ion side product. Big scale industrial methods for the production of simple amines tend in fact to prefer the amination of alcohols as the side product is water. That said, it remains an indispensable tool of the synthetic organic chemist’s toolbox.
Reference Reaction Protocols
Prepare a solution of the amine and the alkyl halide in DMF. Using an excess of either starting material is beneficial (1.5-2 equivalents). Add 1.5 equivalents of the base and 1.5 equivalents of NaI in the case the halide is a bromide or a chloride. Stir at RT for 12-24h. For lower reactivity building blocks, increasing the temperature up to reflux conditions is necessary. At the end of the reaction, quench with water, extract with a suitable organic solvent (e.g. AcOEt) and proceed to the product purification.
Examples

Patent Reference: WO2015088045
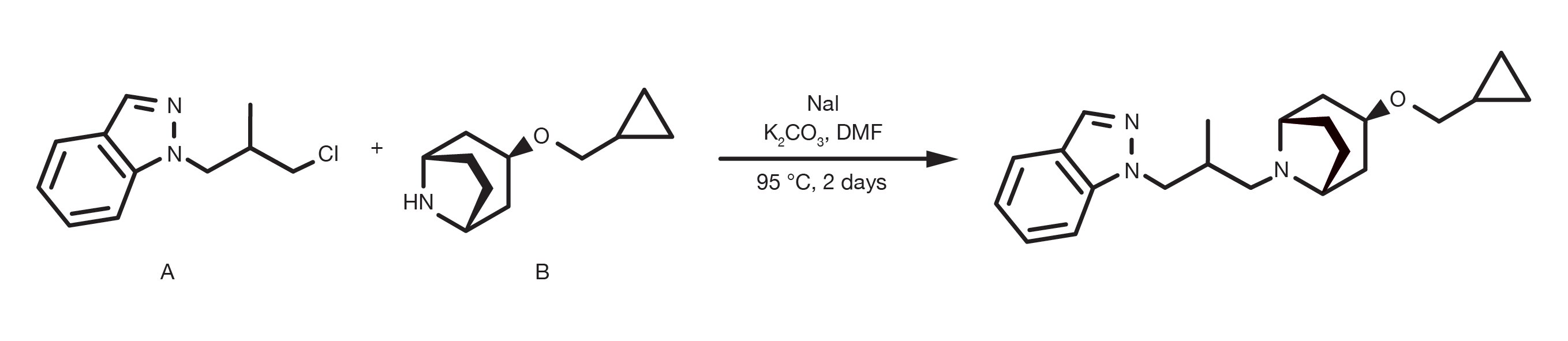
Patent Reference: WO2014152144
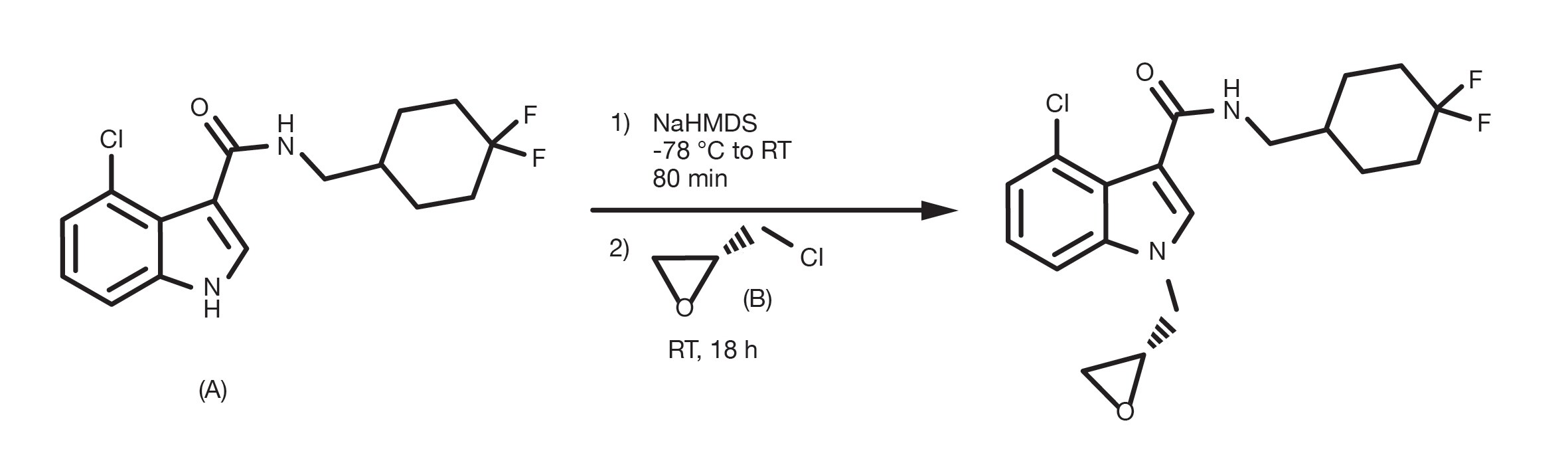
Patent Reference: WO2016100281
Key literature references
- Eller, Karsten; Henkes, Erhard; Rossbacher, Roland; Höke, Hartmut "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry (2000). doi:10.1002/14356007.a02_001
Product Selection
Solvents:
DMF
AcN
Toluene
Acetone
Diethylether
THF
DMSO
EtOH
Basic ingredients / Additives:
K2CO3
Diisopropylethylamine
KOH
CeCl3
1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU)
Sodium Bis(trimethylsilyl)amide
Cs2CO3
NaI
NaH
t-BuOK
t-BuONa
Tetrabutylammonium Iodide
Building blocks:
Variously substituted alkyl bromides (primary and secondary)
Variously substituted alkyl chlorides (primary and secondary)
Variously substituted alkyl iodides (primary and secondary)
Aromatic derivatisation is ok so long as the halide is alkyl!
Variously substituted primary and secondary amines (primary better)
Ammonia
Variously substituted primary and secondary amides
Catalysts/ligands:
--
C-N Coupling
Although the first C-N couplings were reported as early as the 1980’s, it was not until the work of professors Buchwald and Hartwig from 1994 to recent years that Pd-catalysed coupling has become the method of choice for the synthesis of aromatic amines. The use of mild reaction conditions, new generation of air stable, very active catalysts have displaced harsher methods, such as the nucleophilic aromatic substitution, while expanding the substrate scope of the transformation.
The work of Professor Buchwald is consistently among the most cited in the chemical literature and the Buchwald-Hartwig C-N coupling is one of the most ubiquitous reactions in modern organic synthesis, a key step in the production of many biologically active compounds of commercial interest.
The reaction allows the Pd-catalysed coupling of virtually any amine with variously substituted aromatic/heteroaromatic halides or triflates. The reaction happens in the presence of a base and it can be performed in many different solvents, with toluene, THF, dioxane and DME being the most common. Even alcohols, such as tBuOH or t-AmOH (better at high temperatures) are good options.
A typical C-N coupling is run at 50-100C for up to 24h, although full conversions in just a few hours are commonly reported.
The most versatile base for this reaction is arguably NaOt-Bu, although it is often necessary to revert to weaker bases to avoid side reactions with electrophilic functional groups or epimerization of acidic carbons.

The combination of the source of Pd(0) and ligand is the most important practical parameter for a successful C-N coupling. There are many options for the transformation, from old generation metal precursors and ligands, such as Pd(OAc) and BINAP, or dppf, to modern generation pre-formed catalysts, such as generation-4 palladacycles or pi-allyl-Pd complexes. Over the years, the use of more electron-rich chelating phosphines, such as Josiphos and trialkylphosphines has expanded the substrate scope and relaxed the reaction conditions. Among the many available catalytic options, however, only a few have seen extensive practical application. The key parameters for the selection of the catalyst are a combination of ease of use (e.g. air stability), robustness, substrate scope and commercial availability. Modern generation catalysts, based on bulky dialkylbiaryl phosphine ligands pioneered by Buchwald and Hartwig’s research, offer several advantages thanks to their air stability and their exceptional activity and have expanded the scope of the reaction enormously. They now represent the most commonly used ligands in C-N coupling reactions.
Reference Reaction Protocols
An incredibly useful practical guide to identify the best reactions conditions for a Buchwald C-N coupling can be found in Chem. Sci., 2011, 2, 27 (DOI: 10.1039/c0sc00331j). A reference protocol is as follows.
Prepare a toluene solution with the aryl halide and the amine in equimolar amounts, the Pd catalyst (or the Pd precursor and ligand mixture) and 2 equivalents of base (Cs2CO3 or NaOt-Bu). A good starting point for screening conditions is the use of a 5-10% catalyst loading that can be reduced with some optimisation work later on. The reaction is carried out stirring at temperatures between 90 and 140 C, preferably under N2 (mandatory for non air-stable catalysts). Conversion times can be very short, even below 1h, even though 12-24h reaction times are commonly reported.
The use of late-generation catalyst make the reaction set up particularly straightforward, as air stable Pd complexes can be used without a dry-box and the reaction performed in normal laboratory glassware. Previous generation catalysts and in particular the use of active Pd(0) species require some precautions beyond the scope of this text.
Examples

Patent Reference: WO2012112946
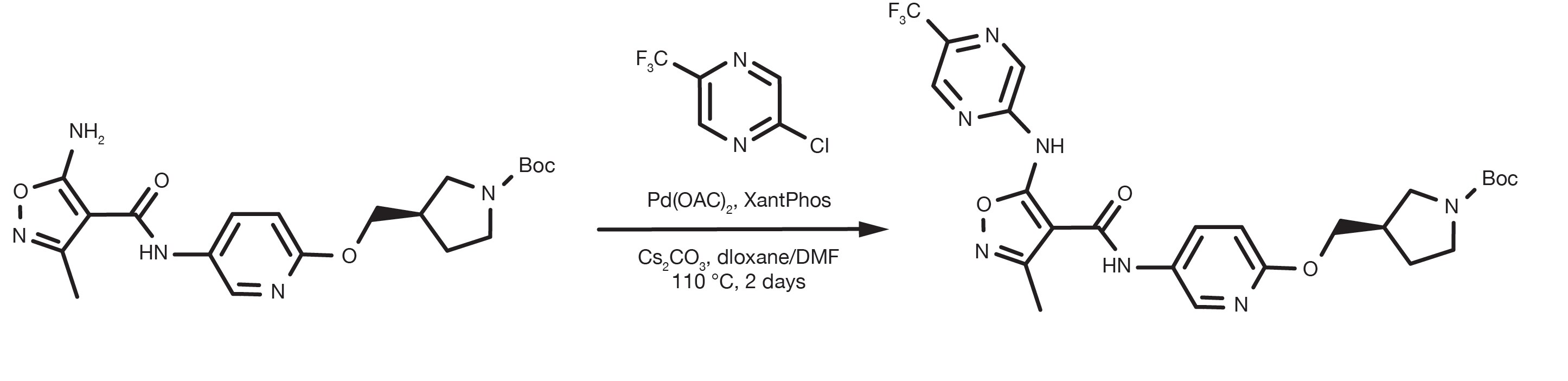
Patent Reference: WO2016012477
Key literature references
- J. Am. Chem. Soc., 1994, 116 (13), pp 5969–5970 DOI: 10.1021/ja00092a058
- J. Am. Chem. Soc., 1994, 116 (17), pp 7901–7902 DOI: 10.1021/ja00096a059
- Journal of Organometallic Chemistry 1999, 576, pp 125-146 https://doi.org/10.1016/S0022-328X(98)01054-7
- Chem. Sci., 2011, 2, 27 DOI: 10.1039/c0sc00331j
- Angewandte Chemie 2008, 47, pp 6338-6361 https://doi.org/10.1002/anie.200800497
Product Selection
Solvents:
Toluene
THF
Dioxane
DMF
H2O
Dimethoxyethane
tBuOH
tert-amyl alcohol
Basic ingredients / Additives:
Toluene
Trifluoroacetic acid
Cs2CO3
Na2CO3
tBuONa
Lithium bis(trimethylsilyl)amide
KOH
K3PO4
Building blocks:
Variously substituted (hetero)aryl chlorides (but also Iodides, bromides)
Variously substituted (hetero)aryl OTf
Primary or secondary alkyl amines
(hetero)aryl amines
Carbamates (as donor of ammonia)
Catalysts/ligands:
Pd-precatalysts
Pd(OAc)2 / [Pd(OAc)2]3
Pd(dba)2
PdCl2(o-Tolyl3)
Pd2(dba)3
PdCl2 (dppf)
Buchwald Palladacycle generation 1
Buchwald Palladacycle gen. 2
Buchwald Palladacycle gen. 3
Buchwald Palladacycle gen. 4
Pre-formed Buchwald Pi-allyl Pd complexes (see JM variants)
Ligands
BINAP
DPPF
Xantphos
Highly hindered dialkylbiaryl phosphine ligands:
DavePhos
JohnPhos
MePhos
XPhos
SPhos
RuPhos
BrettPhos
CPhos
AlPhos
And all their variants where available!

